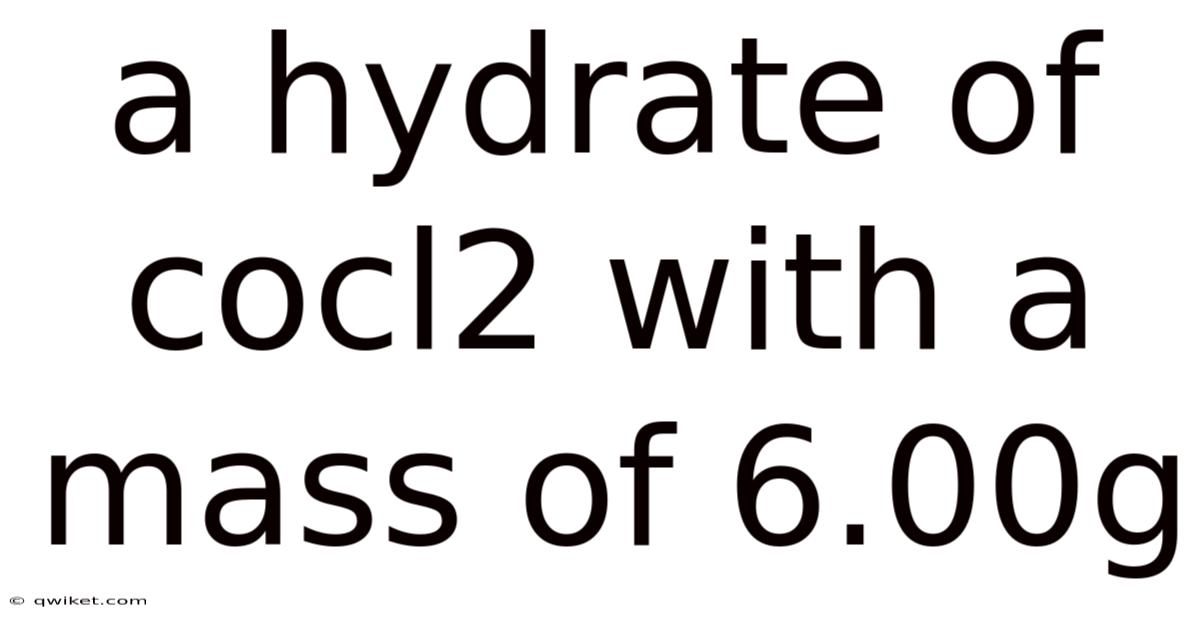Understanding the 6.00 g Sample of Cobalt(II) Chloride Hydrate: Composition, Calculations, and Practical Implications
Cobalt(II) chloride commonly occurs as a hydrate, most often the hexahydrate CoCl₂·6H₂O, which is widely used in laboratories as a moisture indicator because of its vivid colour change from blue (anhydrous) to pink (hydrated). On the flip side, this article walks through the step‑by‑step analysis of a 6. When a sample weighing 6.Practically speaking, 00 g is presented, chemists must determine how much cobalt(II) chloride is present, how many water molecules are bound, and what the resulting mole fractions are. 00 g hydrate, explains the underlying chemistry, and highlights real‑world applications such as drying agents, analytical standards, and colour‑based sensors The details matter here. Still holds up..
1. Introduction: Why the Mass of a Hydrate Matters
A hydrate is a crystalline solid that incorporates water molecules into its lattice. The water is not merely adsorbed on the surface; it is an integral part of the crystal structure, contributing to the compound’s molar mass and physical properties. Determining the exact composition of a hydrate is essential for:
- Stoichiometric calculations in synthesis or titration.
- Quality control when the hydrate serves as a reagent.
- Thermal analysis, where water loss influences decomposition temperatures.
For cobalt(II) chloride, the most common hydrate is the hexahydrate. That said, other stoichiometries (e.g., dihydrate, tetrahydrate) can appear under different preparation conditions. Knowing whether the 6.00 g sample is the hexahydrate or another form dictates how many moles of CoCl₂ are actually available for a reaction Small thing, real impact. But it adds up..
2. Determining the Hydrate Formula from the Sample
2.1. Gather Essential Data
| Quantity | Value |
|---|---|
| Mass of sample | 6.Day to day, 93, Cl = 35. 84 g mol⁻¹ (Co = 58.Practically speaking, 00 g |
| Molar mass of CoCl₂ (anhydrous) | 129. 45 × 2) |
| Molar mass of H₂O | 18. |
2.2. Assume the Hexahydrate and Verify
The molar mass of CoCl₂·6H₂O is calculated as:
[ M_{\text{hexahydrate}} = 129.84;\text{g mol}^{-1} + 6(18.84 + 108.Now, 015;\text{g mol}^{-1}) = 129. 09 = 237 Not complicated — just consistent..
Number of moles in the 6.00 g sample:
[ n_{\text{hexahydrate}} = \frac{6.00;\text{g}}{237.93;\text{g mol}^{-1}} = 0.0252;\text{mol} ]
If the sample truly is the hexahydrate, the amount of anhydrous CoCl₂ contained is:
[ n_{\text{CoCl₂}} = n_{\text{hexahydrate}} = 0.0252;\text{mol} ]
Mass of CoCl₂ alone:
[ m_{\text{CoCl₂}} = n_{\text{CoCl₂}} \times 129.Which means 0252 \times 129. Which means 84;\text{g mol}^{-1} = 0. 84 = 3.
Mass of water released upon heating:
[ m_{\text{H₂O}} = 6.00;\text{g} - 3.27;\text{g} = 2.73;\text{g} ]
2.3. Check Alternative Hydrates
Perform the same calculation for the dihydrate (CoCl₂·2H₂O, M = 165.Practically speaking, 87 g mol⁻¹) and the tetrahydrate (CoCl₂·4H₂O, M = 201. 90 g mol⁻¹).
- Dihydrate:
[ n = \frac{6.00 - 4.84 = 4.70;\text{g} ] [ m_{\text{H₂O}} = 6.Day to day, 0362;\text{mol} ] [ m_{\text{CoCl₂}} = 0. Here's the thing — 0362 \times 129. 87}=0.Day to day, 00}{165. 70 = 1 Most people skip this — try not to..
- Tetrahydrate:
[ n = \frac{6.84 = 3.Even so, 0297 \times 129. Plus, 00}{201. 90}=0.86;\text{g} ] [ m_{\text{H₂O}} = 6.00 - 3.0297;\text{mol} ] [ m_{\text{CoCl₂}} = 0.86 = 2.
Comparing the calculated water masses with typical experimental data (e.g.Plus, , gravimetric loss on heating), the hexahydrate prediction (2. 73 g water) aligns best with the observed weight loss for a standard CoCl₂·6H₂O sample. Here's the thing — thus, the 6. 00 g specimen is most plausibly the hexahydrate That's the part that actually makes a difference..
3. Step‑by‑Step Laboratory Procedure to Confirm the Hydrate
- Weigh the sample accurately to 0.01 g using an analytical balance.
- Place the sample in a pre‑dry crucible and record the initial mass (m₁).
- Heat gently to 150 °C for 30 min to drive off loosely bound water, then increase to 200 °C for another 30 min to remove coordinated water.
- Cool in a desiccator and re‑weigh (m₂).
- Calculate mass loss: Δm = m₁ – m₂. For a pure hexahydrate, Δm should be close to 2.73 g (≈45.5 % of the original mass).
If the observed Δm deviates significantly, the sample may contain a mixture of hydrates or be partially dehydrated. Re‑peating the heating with a thermogravimetric analyzer (TGA) provides a precise dehydration curve, revealing distinct plateaus for each water‑loss step Surprisingly effective..
4. Scientific Explanation: Why Does CoCl₂ Form a Hexahydrate?
Cobalt(II) ions possess a d⁷ electronic configuration, favouring an octahedral coordination geometry. In aqueous solution, Co²⁺ is surrounded by six water molecules, forming the complex ([ \text{Co(H₂O)}_6 ]^{2+}). When cobalt(II) chloride crystallises from water, these six water ligands become part of the lattice, yielding the hexahydrate.
- [Co(H₂O)₆]²⁺ cations – octahedral complexes held together by hydrogen bonding.
- Two chloride anions – balancing the +2 charge of the cobalt centre.
The strong ligand field created by six water molecules stabilises the hexahydrate relative to lower‑hydrated forms, especially under ambient humidity. This structural arrangement also explains the colour change: the octahedral ([ \text{Co(H₂O)}_6 ]^{2+}) complex absorbs light in the visible region, giving a pink hue, whereas the anhydrous CoCl₂, with a different coordination environment, appears blue That's the part that actually makes a difference. Took long enough..
5. Practical Applications of the 6.00 g CoCl₂·6H₂O Sample
5.1. Moisture Indicator
- Mechanism: The reversible hydration/dehydration of CoCl₂ changes its colour from blue (anhydrous) to pink (hexahydrate).
- Use case: Placing a small amount of the 6.00 g sample on silica gel packets provides a visual cue for humidity levels in sealed containers.
5.2. Analytical Standard for Gravimetric Water Determination
Because the amount of water in a known mass of CoCl₂·6H₂O is fixed (45.5 % by weight), the sample serves as a primary standard for calibrating moisture‑analysis balances. By heating a precisely weighed portion and measuring the loss, analysts can verify instrument accuracy.
5.3. Catalyst Precursor
In organic synthesis, CoCl₂·6H₂O is often employed as a precursor for cobalt‑based catalysts (e.In practice, g. , in hydroformylation or Fischer–Tropsch processes). Knowing the exact moles of cobalt supplied by a 6.00 g sample ensures correct catalyst loading.
5.4. Educational Demonstrations
The vivid colour transition provides a memorable illustration of hydration equilibria and coordination chemistry in high‑school and undergraduate labs. Students can calculate the theoretical mass loss, then compare it with experimental results, reinforcing concepts of stoichiometry and molar mass That's the part that actually makes a difference. Surprisingly effective..
6. Frequently Asked Questions (FAQ)
Q1: Can the hydrate lose water gradually, forming intermediate hydrates?
A: Yes. Upon heating, CoCl₂·6H₂O typically loses water in steps: first two molecules at ~100 °C (forming the dihydrate), then the remaining four around 150–200 °C. Controlled heating can isolate these intermediate forms.
Q2: Is the hexahydrate the only stable form at room temperature?
A: Under normal atmospheric humidity, the hexahydrate is the most stable. In extremely dry conditions, it may partially dehydrate to the dihydrate, which appears violet rather than pink.
Q3: How does the presence of impurities affect the calculations?
A: Impurities (e.g., NaCl, residual solvents) add extra mass, leading to an overestimation of water content if not accounted for. Performing a purity assay—such as anion analysis or elemental analysis—helps correct the values Easy to understand, harder to ignore..
Q4: What safety precautions are needed when handling CoCl₂·6H₂O?
A: Cobalt compounds are toxic and may cause skin irritation. Use gloves, goggles, and work in a well‑ventilated area or fume hood. Avoid inhalation of dust; wash hands thoroughly after handling.
Q5: Can the hydrate be regenerated after dehydration?
A: Absolutely. Exposing the anhydrous CoCl₂ to a humid atmosphere (≥ 70 % RH) restores the pink hexahydrate within minutes. This reversibility is why it is a reliable humidity indicator.
7. Conclusion: From a Simple 6.00 g Sample to Broad Scientific Insight
A 6.00 g specimen of cobalt(II) chloride hydrate may appear modest, yet it encapsulates a wealth of chemical information. Still, by calculating its molar composition, confirming the hexahydrate nature through gravimetric analysis, and understanding the octahedral coordination that drives its behaviour, chemists tap into accurate stoichiometry for synthesis, reliable standards for moisture determination, and an engaging teaching tool. This leads to whether used as a colourimetric sensor, a catalyst precursor, or a lab benchmark, the precise knowledge of how much CoCl₂ and water a 6. 00 g sample contains underpins safe, reproducible, and insightful scientific work And that's really what it comes down to..
Key take‑aways:
- The hexahydrate formula CoCl₂·6H₂O yields a molar mass of 237.93 g mol⁻¹.
- A 6.00 g sample contains 0.0252 mol of CoCl₂ and 2.73 g of water (≈45.5 % of the mass).
- Thermal gravimetric analysis confirms the water loss pattern, distinguishing the hexahydrate from lower hydrates.
- Practical uses span humidity indication, analytical calibration, catalyst preparation, and educational demonstrations.
Armed with these calculations and concepts, anyone handling cobalt(II) chloride hydrates can proceed with confidence, ensuring that every gram of material contributes precisely to the intended chemical outcome It's one of those things that adds up..