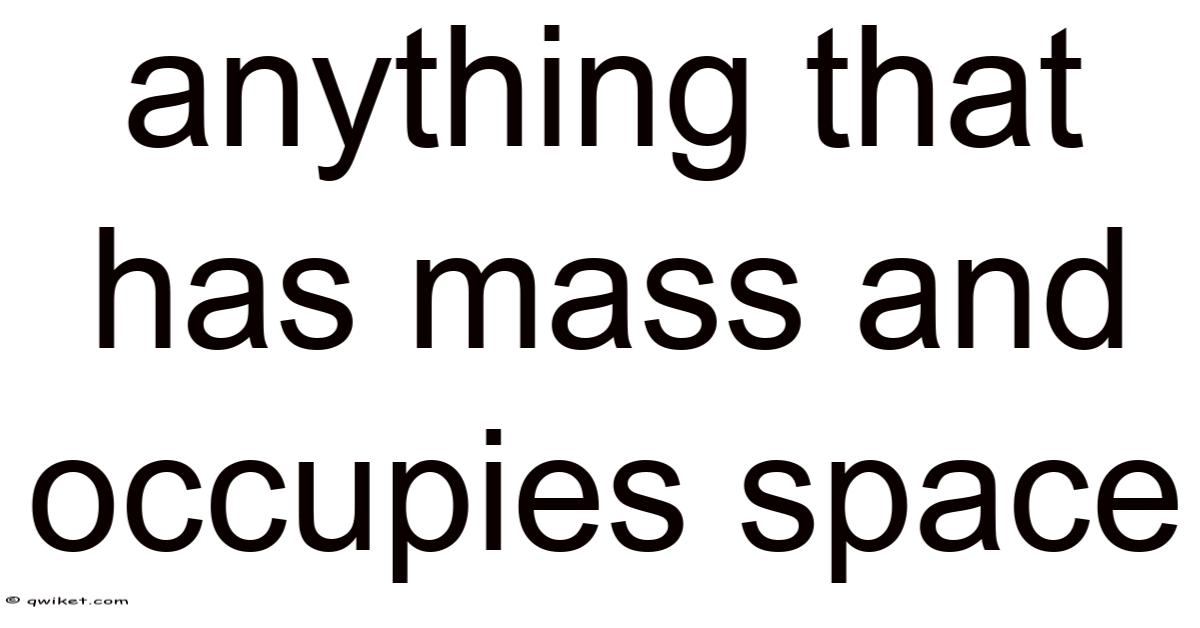Anything that has mass and occupies space is the very definition of matter, the fundamental substance that makes up the universe we experience. Still, from the tiniest subatomic particles to the colossal galaxy clusters, every tangible entity conforms to this simple yet profound rule: it possesses mass and takes up volume. Understanding what matter is, how it behaves, and why it matters (pun intended) provides a gateway to grasping physics, chemistry, biology, and even everyday phenomena. This article explores the nature of matter, its various states, the forces that govern it, and the intriguing exceptions that challenge our classical intuition Not complicated — just consistent..
Introduction: Why Matter Matters
When you pick up a coffee mug, feel the warmth of the sun on your skin, or watch a raindrop splash into a puddle, you are interacting with matter. The phrase “anything that has mass and occupies space” may sound like a textbook definition, but it encapsulates the essence of the material world. Recognizing matter’s properties allows us to predict how objects will move, react, and transform—a cornerstone of scientific inquiry and technological advancement But it adds up..
The Core Definition of Matter
- Mass: A measure of an object’s resistance to acceleration (inertia) and a source of gravitational attraction. It is quantified in kilograms (kg) in the International System of Units (SI).
- Volume: The three‑dimensional space an object occupies, measured in cubic meters (m³) or liters (L).
When both criteria are met, the entity is classified as matter. This excludes pure energy (photons) and abstract concepts (numbers, ideas), which lack rest mass and spatial extension.
Forms of Matter: The Classical States
1. Solid
Solids retain a fixed shape and volume because their constituent particles (atoms, ions, or molecules) are tightly packed in a regular lattice. The strong intermolecular forces restrict movement to vibrations around equilibrium positions Turns out it matters..
- Examples: Diamond, iron, ice.
- Key properties: High density, definite shape, low compressibility.
2. Liquid
Liquids have a definite volume but no fixed shape; they flow to adopt the contour of their container. The particles are less tightly bound than in solids, allowing them to slide past one another while remaining cohesive That's the part that actually makes a difference..
- Examples: Water, mercury, ethanol.
- Key properties: Moderate density, surface tension, ability to dissolve many solids (solvent role).
3. Gas
Gases possess neither fixed shape nor fixed volume. Here's the thing — their particles move freely and are far apart, filling any container uniformly. Intermolecular forces are negligible compared to kinetic energy.
- Examples: Oxygen, carbon dioxide, steam.
- Key properties: Low density, high compressibility, obey ideal gas law (PV = nRT) under many conditions.
4. Plasma
Often called the “fourth state of matter,” plasma consists of ionized gas with free electrons and positively charged ions. It conducts electricity and responds strongly to magnetic fields.
- Examples: Sun’s core, neon signs, lightning.
- Key properties: High temperature, collective behavior, emits light.
5. Bose‑Einstein Condensate (BEC)
At temperatures near absolute zero, certain bosonic atoms occupy the same quantum ground state, behaving as a single quantum entity. BECs demonstrate macroscopic quantum phenomena Easy to understand, harder to ignore..
- Examples: Rubidium‑87 condensates created in labs.
- Key properties: Superfluidity, coherence, extremely low entropy.
The Building Blocks of Matter
Atoms: The Fundamental Units
An atom comprises a nucleus (protons and neutrons) surrounded by electrons in orbitals. The mass of an atom is almost entirely due to the nucleus, while the volume is defined by the electron cloud’s spatial extent.
- Protons: Positive charge, mass ≈ 1.67 × 10⁻²⁷ kg.
- Neutrons: Neutral charge, similar mass to protons.
- Electrons: Negative charge, mass ≈ 9.11 × 10⁻³¹ kg (≈ 1/1836 of a proton).
Molecules and Compounds
When atoms bond chemically, they form molecules—larger entities that still satisfy the mass‑and‑space criteria. Chemical bonds (covalent, ionic, metallic) determine the arrangement and properties of matter at the macroscopic level.
Subatomic Particles Beyond Atoms
- Quarks: Constituents of protons and neutrons; they possess mass and occupy space within nucleons.
- Leptons (e.g., electrons, muons): Have mass and occupy space, though their “size” is defined by quantum probability distributions rather than a hard boundary.
Forces Governing Matter
Gravitational Force
All matter exerts an attractive force on other matter proportional to their masses (Newton’s law of universal gravitation). This force shapes planetary orbits, star formation, and the large‑scale structure of the cosmos.
Electromagnetic Force
Charged particles attract or repel each other, governing chemical bonding, electrical conductivity, and the behavior of plasma. The electromagnetic interaction is responsible for the volume of matter, as electron clouds repel each other, preventing particles from collapsing into a point.
Strong Nuclear Force
Acts over femtometer distances, binding protons and neutrons within the nucleus. Without it, the repulsive electromagnetic force between protons would cause nuclei to disintegrate Easy to understand, harder to ignore..
Weak Nuclear Force
Responsible for certain types of radioactive decay (beta decay), it changes the identity of particles, influencing the stability and transformation of matter.
Matter vs. Energy: The Relativistic Connection
Einstein’s famous equation, E = mc², reveals that mass and energy are interchangeable. Even so, photons do not occupy space in the classical sense; they are described by wavefunctions that spread out, not by a defined volume. While photons (light particles) have zero rest mass, they carry energy and momentum, allowing them to exert pressure (radiation pressure) and influence matter. So g. Think about it: thus, the strict definition of matter excludes pure energy, even though mass can be converted to energy (e. , nuclear reactions) Most people skip this — try not to..
Short version: it depends. Long version — keep reading.
Exotic Forms and Edge Cases
Dark Matter
Astrophysical observations (galaxy rotation curves, gravitational lensing) imply a massive, invisible component that does not emit, absorb, or reflect light. Worth adding: dark matter has mass and exerts gravitational influence, yet its composition remains unknown. It satisfies the mass‑and‑space definition only insofar as it contributes to the overall mass density of the universe; its “space” is inferred indirectly.
Antimatter
Antiparticles possess the same mass as their matter counterparts but opposite charge. When matter and antimatter meet, they annihilate, converting mass into pure energy (photons). Antimatter is still matter under the definition because it has mass and occupies space until annihilation occurs Most people skip this — try not to..
Neutrinos
These elusive particles have a tiny, nonzero mass and travel near light speed. Their interaction cross‑section with ordinary matter is minuscule, making them effectively ghost‑like, yet they still fulfill the criteria of mass and spatial presence.
Measuring Mass and Volume
- Mass: Determined using balances (comparative, spring, electronic) that compare an unknown mass to known standards.
- Volume: Measured directly for regular shapes (geometric formulas) or via displacement methods (Archimedes’ principle) for irregular objects. For gases, volume is inferred from pressure, temperature, and amount of substance using the ideal gas law.
Everyday Applications of Matter’s Principles
- Engineering Materials: Selecting metals, polymers, or composites based on density, strength, and thermal expansion—properties rooted in mass and volume.
- Pharmaceuticals: Dosing depends on the mass of active ingredients; solubility relates to how molecules occupy space in solvents.
- Environmental Science: Tracking pollutant mass balances and dispersal volumes informs remediation strategies.
- Astronomy: Estimating stellar masses and planetary volumes enables calculations of gravitational forces and orbital dynamics.
Frequently Asked Questions (FAQ)
Q1: Can something have mass but no volume?
A: In classical physics, mass always implies some spatial extent. Quantum mechanics introduces point‑like particles (e.g., electrons) that are treated as having no classical radius, yet their probability clouds define an effective volume.
Q2: Does vacuum contain matter?
A: A perfect vacuum lacks matter, but quantum field theory predicts fleeting particle‑antiparticle pairs (virtual particles) that momentarily possess mass and occupy space, though they do not constitute persistent matter Simple, but easy to overlook..
Q3: How does temperature affect the volume of matter?
A: Raising temperature generally increases kinetic energy, causing particles to move apart, expanding the volume of gases (Charles’s law) and, to a lesser extent, liquids and solids (thermal expansion).
Q4: Why do we consider plasma a state of matter if it’s ionized gas?
A: Ionization separates electrons from nuclei, creating a mixture of charged particles that exhibit collective behavior distinct from neutral gases, justifying its classification as a separate state That's the part that actually makes a difference..
Q5: Is dark energy considered matter?
A: No. Dark energy is a property of space itself that drives the accelerated expansion of the universe. It lacks mass and does not occupy space in the conventional sense Which is the point..
Conclusion: The Central Role of Matter in Science and Life
Everything we touch, see, and manipulate conforms to the simple rule: anything that has mass and occupies space is matter. Worth adding: from the microscopic dance of electrons in a semiconductor to the gravitational ballet of galaxies, matter’s properties dictate the behavior of the physical world. Worth adding: recognizing the various states, the forces that bind particles, and the exceptional cases like dark matter or antimatter enriches our comprehension of nature’s complexity. Day to day, as we continue to probe deeper—through particle accelerators, space telescopes, and quantum experiments—we constantly refine our definition of what constitutes matter, reminding us that even the most fundamental concepts evolve with discovery. Understanding matter is not just an academic exercise; it is the foundation upon which technology, medicine, and environmental stewardship are built, shaping the future of humanity.