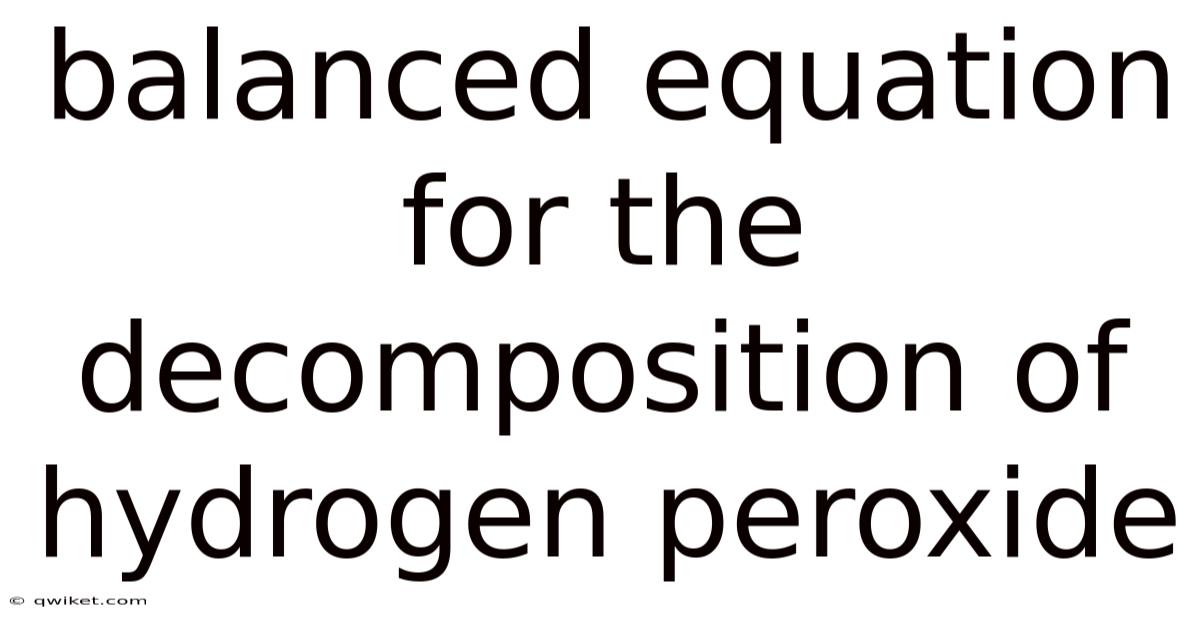The decomposition of hydrogen peroxide presents a fascinating intersection of chemistry, environmental science, and practical applications. While often overlooked in casual discourse, this reaction serves as a critical lens through which we examine the dynamic interplay between molecular structure and observable outcomes. That's why hydrogen peroxide (H₂O₂), a compound notorious for its dual roles as both a powerful oxidizing agent and a potential environmental contaminant, finds itself at the center of numerous scientific investigations. Its decomposition, though seemingly simple on the surface, reveals complexities rooted in the fundamental principles governing chemical equilibrium and energy transfer. This process not only illuminates the intricacies of molecular behavior but also underscores the importance of precise mathematical representation in managing real-world phenomena. But the very act of balancing the equation governing its transformation underscores the necessity of meticulous attention to detail, ensuring that the nuanced dance of atoms and molecules is accurately captured. Consider this: such precision is essential, as even minor deviations can lead to unintended consequences, whether in laboratory settings or natural ecosystems. Now, in this context, understanding the balanced equation becomes more than a technical exercise; it transforms into a foundational tool for grasping the underlying dynamics that govern the process. It serves as a bridge connecting theoretical knowledge to practical implementation, offering insights that extend far beyond the confines of a single reaction. Here, the equation stands as a testament to the precision required when dealing with substances that possess both utility and potential hazards, highlighting the delicate balance between science and application. Day to day, such equilibrium is not merely a mathematical construct but a practical consideration that demands careful stewardship, reinforcing the value placed on accuracy in scientific endeavors. The implications of this balance ripple through various fields, from industrial chemistry to ecological studies, where the outcomes of H₂O₂ decomposition can significantly influence environmental stability and human health. That said, thus, the study of this reaction transcends its immediate context, positioning it as a cornerstone in the broader narrative of chemical processes that shape our world. The foundational equation provides the scaffold upon which deeper explorations can be built, allowing researchers and educators to build upon its principles while addressing the multifaceted challenges associated with its application. Such foundational knowledge forms the basis for further inquiry, making it a critical element in advancing our collective understanding of chemical reactivity and its real-world manifestations.
The balanced equation for the decomposition of hydrogen peroxide is 2H₂O₂ → 2H₂O + O₂. This stoichiometric relationship is not merely a numerical arrangement but a reflection of the conservation laws at play—specifically, the preservation of mass and charge. Here's the thing — on the left side, two molecules of hydrogen peroxide contribute a total of four hydrogen atoms and four oxygen atoms, yielding two water molecules (each with two hydrogens and one oxygen) and one molecule of oxygen gas (with two oxygen atoms). On the right, the two water molecules account for four hydrogens and two oxygens, while the oxygen gas provides the remaining two oxygen atoms, balancing the total. This balance ensures that no atoms are created or destroyed, a principle central to all chemical reactions. Yet, despite its apparent simplicity, the equation’s derivation often requires careful scrutiny to avoid oversight. Still, for instance, one might mistakenly overlook the stoichiometric coefficients, leading to miscalculations that could compromise experimental validity. The process involves meticulous verification of each element’s count, ensuring that the equation accurately mirrors the physical transformation occurring. Such precision is crucial when applying this knowledge in practical scenarios, where even minor errors could result in inefficiencies or safety risks. What's more, the equation serves as a template for analogous reactions, allowing chemists to extrapolate solutions based on established frameworks.
The balanced equation for the decomposition of hydrogen peroxide is 2H₂O₂ → 2H₂O + O₂. This stoichiometric relationship is not merely a numerical arrangement but a reflection of the conservation laws at play—specifically, the preservation of mass and charge. On the left side, two molecules of hydrogen peroxide contribute a total of four hydrogen atoms and four oxygen atoms, yielding two water molecules (each with two hydrogens and one oxygen) and one molecule of oxygen gas (with two oxygen atoms). On the right, the two water molecules account for four hydrogens and two oxygens, while the oxygen gas provides the remaining two oxygen atoms, balancing the total. This balance ensures that no atoms are created or destroyed, a principle central to all chemical reactions. Still, yet, despite its apparent simplicity, the equation’s derivation often requires careful scrutiny to avoid oversight. Take this case: one might mistakenly overlook the stoichiometric coefficients, leading to miscalculations that could compromise experimental validity. On top of that, the process involves meticulous verification of each element’s count, ensuring that the equation accurately mirrors the physical transformation occurring. Such precision is crucial when applying this knowledge in practical scenarios, where even minor errors could result in inefficiencies or safety risks. Adding to this, the equation serves as a template for analogous reactions, allowing chemists to extrapolate solutions based on established frameworks. This foundational understanding not only aids in predicting reaction outcomes but also informs the design of experiments and industrial processes, where the controlled decomposition of hydrogen peroxide is leveraged for applications ranging from disinfection to propulsion systems. The interplay between theoretical principles and practical applications underscores the enduring relevance of this seemingly straightforward reaction, highlighting its role as a bridge between abstract chemical concepts and tangible real-world solutions.
foundational understanding not only aids in predicting reaction outcomes but also informs the design of experiments and industrial processes, where the controlled decomposition of hydrogen peroxide is leveraged for applications ranging from disinfection to propulsion systems. The interplay between theoretical principles and practical applications underscores the enduring relevance of this seemingly straightforward reaction, highlighting its role as a bridge between abstract chemical concepts and tangible real-world solutions. By mastering the balance of such equations, chemists confirm that their work remains both scientifically rigorous and practically effective, reinforcing the critical importance of precision in the field.