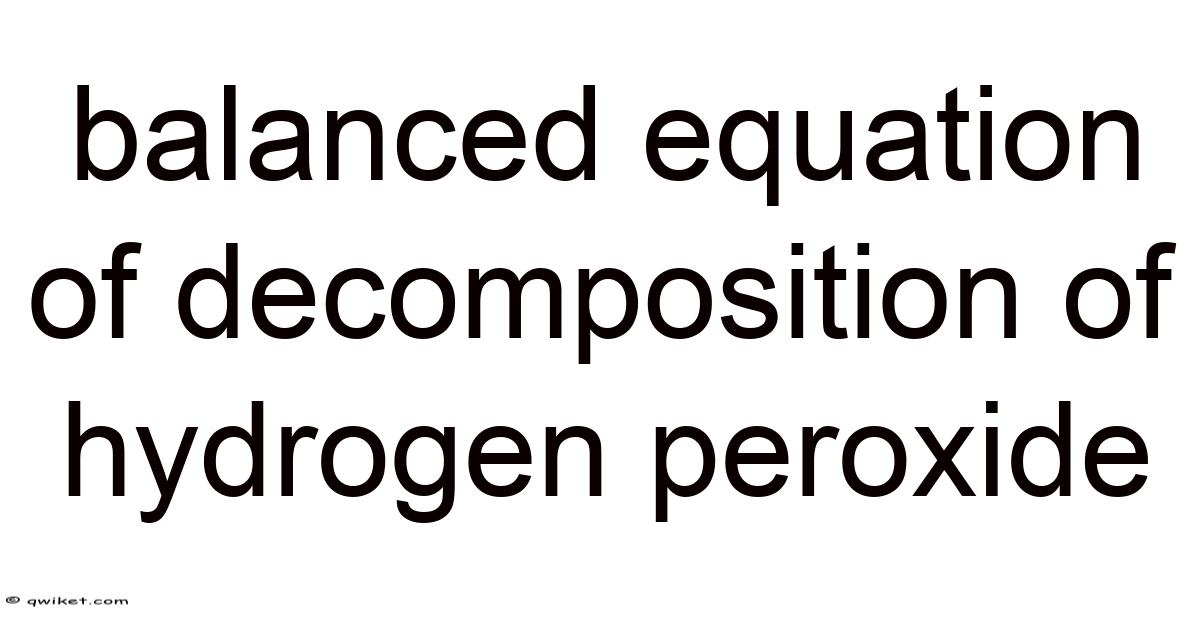Balanced Equation of Decomposition of Hydrogen Peroxide
The decomposition of hydrogen peroxide is one of the most commonly studied chemical reactions in chemistry education and laboratory practice. That said, hydrogen peroxide, a familiar household antiseptic and industrial bleaching agent, naturally breaks down into water and oxygen over time. So understanding the balanced equation for this reaction is essential for students, educators, and professionals who work with oxidizing agents. This article provides a comprehensive look at the balanced chemical equation for the decomposition of hydrogen peroxide, the science behind the reaction, the factors that influence it, and its real-world significance Turns out it matters..
What Is Hydrogen Peroxide?
Hydrogen peroxide is a chemical compound with the molecular formula H₂O₂. It is a pale blue liquid that appears colorless in dilute solutions. Structurally, it is very similar to water (H₂O), but it contains an extra oxygen atom, making it a powerful oxidizing agent. This extra oxygen is what makes hydrogen peroxide useful in a wide range of applications, from wound cleaning and hair bleaching to rocket propulsion and wastewater treatment.
In its pure form, hydrogen peroxide is relatively unstable. It tends to break apart spontaneously, which is why commercial hydrogen peroxide solutions are typically stored in dark, cool environments to slow down this natural breakdown process The details matter here..
What Is Decomposition of Hydrogen Peroxide?
Decomposition is a type of chemical reaction in which a single compound breaks down into two or more simpler substances. In the case of hydrogen peroxide, the compound splits into water (H₂O) and oxygen gas (O₂). This reaction can occur on its own at a very slow rate, but it is significantly accelerated in the presence of a catalyst That's the whole idea..
A catalyst is a substance that speeds up a chemical reaction without being consumed in the process. Common catalysts used for the decomposition of hydrogen peroxide include manganese dioxide (MnO₂), potassium iodide (KI), and the biological enzyme catalase, which is found in living cells Small thing, real impact. Surprisingly effective..
The Balanced Chemical Equation
The balanced equation for the decomposition of hydrogen peroxide is as follows:
2 H₂O₂ (aq) → 2 H₂O (l) + O₂ (g)
Let us break this equation down:
- Reactant: Two molecules of hydrogen peroxide (H₂O₂) in aqueous solution.
- Products: Two molecules of water (H₂O) in liquid form and one molecule of oxygen gas (O₂).
Why Is This Equation Balanced?
A balanced chemical equation must have the same number of atoms of each element on both sides of the equation. Let us verify:
| Element | Reactant Side (2 H₂O₂) | Product Side (2 H₂O + O₂) |
|---|---|---|
| Hydrogen (H) | 2 × 2 = 4 atoms | 2 × 2 = 4 atoms |
| Oxygen (O) | 2 × 2 = 4 atoms | (2 × 1) + 2 = 4 atoms |
Counterintuitive, but true.
As shown in the table, both hydrogen and oxygen atoms are equal on each side, confirming that the equation is properly balanced. This balance reflects the law of conservation of mass, which states that matter cannot be created or destroyed in a chemical reaction.
The Role of Catalysts in the Decomposition Reaction
Without a catalyst, the decomposition of hydrogen peroxide proceeds extremely slowly at room temperature. Adding a catalyst dramatically increases the rate of the reaction. Here are some of the most commonly used catalysts:
-
Manganese Dioxide (MnO₂): This is the most popular catalyst used in school laboratory demonstrations. When a small amount of MnO₂ powder is added to hydrogen peroxide solution, vigorous bubbling of oxygen gas is immediately observed.
-
Potassium Iodide (KI): Often used in the famous "elephant toothpaste" experiment, potassium iodide provides iodide ions (I⁻) that act as a catalyst, rapidly decomposing hydrogen peroxide and producing a dramatic foam of oxygen.
-
Catalase Enzyme: This is a biological catalyst found in the cells of nearly all living organisms. Catalase is extraordinarily efficient, capable of converting millions of hydrogen peroxide molecules into water and oxygen per second. This is vital because hydrogen peroxide is a toxic byproduct of cellular metabolism, and living cells must break it down quickly to prevent damage Not complicated — just consistent..
-
Iron (III) Chloride (FeCl₃): Another inorganic catalyst that can be used to accelerate the decomposition reaction in laboratory settings.
The role of a catalyst is to lower the activation energy of the reaction, meaning that fewer molecular collisions are needed to initiate the breakdown of hydrogen peroxide. Importantly, the catalyst does not appear in the overall balanced equation because it is not consumed during the reaction.
People argue about this. Here's where I land on it Simple, but easy to overlook..
Scientific Explanation of the Reaction Mechanism
At the molecular level, the decomposition of hydrogen peroxide involves the breaking of the O–O bond within the peroxide molecule. This bond is relatively weak compared to other chemical bonds, which is why hydrogen peroxide is inherently unstable.
The reaction can be summarized in the following steps:
- The catalyst provides an alternative reaction pathway with a lower activation energy.
- Hydrogen peroxide molecules adsorb onto the surface of the catalyst (in the case of solid catalysts like MnO₂).
- The O–O bond breaks, releasing oxygen atoms that combine to form O₂ gas.
- The remaining hydrogen atoms combine with oxygen atoms to form water molecules.
- The products (water and oxygen) desorb from the catalyst surface, leaving the catalyst unchanged and ready to support another reaction.
This process is classified as a disproportionation reaction (also known as a dismutation reaction), because the oxygen in hydrogen peroxide (which has an oxidation state of –1) is simultaneously oxidized to form O₂ (oxidation state 0) and reduced to form H₂O (oxidation state –2).
Factors Affecting the Rate of Decomposition
Several factors influence how quickly hydrogen peroxide decomposes:
- Concentration of Hydrogen Peroxide: Higher concentrations decompose more vigorously, producing oxygen at a faster rate.
- Temperature: Increasing the temperature provides molecules with more kinetic energy, speeding up the reaction.
- Presence and Amount of Catalyst: More catalyst or a more effective catalyst will increase the rate of decomposition.
- pH of the Solution: The decomposition rate is affected by the acidity or alkalinity of the solution. Alkaline conditions tend to accelerate decomposition.
- Presence of Stabilizers: Commercial hydrogen peroxide solutions often contain stabilizers such as sodium stannate or phosphoric acid to slow down decomposition and extend shelf life.
Practical Applications of Hydrogen Peroxide Decomposition
The decomposition of hydrogen peroxide is not just an academic exercise. It has numerous practical applications across various industries:
-
Medical and Healthcare: Hydrogen peroxide is widely used as a disinfectant and antiseptic. When it contacts tissues, the enzyme catalase in blood and cells rapidly decomposes it, releasing oxygen that helps clean wounds by mechanically removing debris But it adds up..
-
Environmental Treatment: In wastewater treatment, hydrogen peroxide
is employed as an oxidizing agent to break down organic pollutants, toxic compounds, and hazardous chemicals that conventional treatment methods cannot fully remove. The oxygen released during decomposition also helps replenish dissolved oxygen levels in aquatic systems, promoting the recovery of degraded ecosystems.
-
Rocket Propulsion: One of the most dramatic applications is in monopropellant and bipropellant rocket engines. The rapid catalytic decomposition of high-concentration hydrogen peroxide generates a large volume of hot steam and oxygen gas, which expands through a nozzle to produce thrust. Early experimental rocket engines, including those developed by the German V-2 program, relied on this principle.
-
Food and Beverage Industry: In the food industry, hydrogen peroxide is used to bleach and sterilize packaging materials, equipment surfaces, and certain food products. Controlled decomposition ensures that no harmful residues remain after treatment.
-
Mining and Metallurgy: The decomposition reaction is harnessed in heap leaching operations, where the oxygen produced assists in the oxidation of metal sulfide ores, facilitating the extraction of metals such as copper, gold, and uranium Most people skip this — try not to..
-
Laboratory and Analytical Chemistry: Researchers routinely use the decomposition of hydrogen peroxide as a model reaction for studying catalytic mechanisms, surface chemistry, and reaction kinetics. Its simplicity and well-characterized behavior make it an ideal system for teaching and research.
Safety Considerations
Despite its many uses, hydrogen peroxide decomposition poses certain hazards that must be managed carefully. Rapid decomposition can lead to a violent release of oxygen gas, causing pressure buildup in closed containers and the risk of explosion. Concentrated solutions can also cause severe chemical burns to skin and eyes. Proper storage in cool, well-ventilated areas away from organic materials and metal contaminants is essential. When handling higher concentrations, appropriate personal protective equipment—including gloves, goggles, and lab coats—should always be worn.
Conclusion
The decomposition of hydrogen peroxide is a fundamental chemical process that illustrates the interplay between molecular stability, catalytic action, and reaction thermodynamics. From the weak O–O bond that makes the molecule inherently reactive to the diverse range of catalysts that can accelerate its breakdown, this reaction serves as a cornerstone example in chemistry education and industrial practice alike. Its broad applicability—in medicine, environmental remediation, aerospace engineering, food safety, and mining—underscores how a seemingly simple disproportionation reaction can have far-reaching consequences. Understanding the factors that govern its rate and the conditions that promote or inhibit decomposition not only enhances our theoretical grasp of chemical kinetics but also enables safer, more efficient, and more sustainable use of hydrogen peroxide across numerous fields.