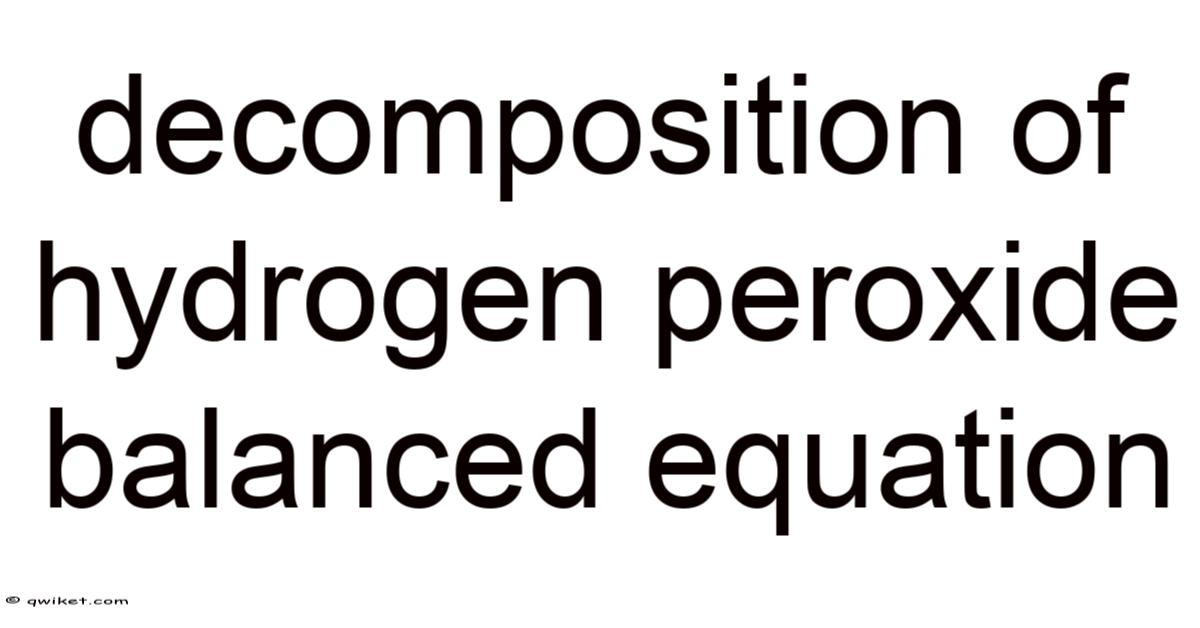Understanding the decomposition of hydrogen peroxide is essential for grasping various chemical processes and reactions. This process involves breaking down hydrogen peroxide into water and oxygen, a reaction that is both fascinating and crucial in numerous applications. By exploring the balanced equation for this decomposition, we can delve deeper into the science behind it and its practical implications Worth knowing..
When we talk about the decomposition of hydrogen peroxide, we refer to the chemical reaction where hydrogen peroxide (H₂O₂) transforms into water (H₂O) and oxygen gas (O₂). This reaction is not only significant in laboratory settings but also plays a vital role in everyday processes, from water treatment to the production of cleaning agents. The balanced equation for this reaction helps us understand the precise amounts of reactants and products involved, ensuring clarity and accuracy in scientific communication Most people skip this — try not to..
The balanced equation for the decomposition of hydrogen peroxide is as follows:
2 H₂O₂ → 2 H₂O + O₂
This equation clearly shows that two molecules of hydrogen peroxide react to produce two molecules of water and one molecule of oxygen gas. The importance of this reaction lies in its ability to release oxygen, which is essential for various biological processes and industrial applications.
To grasp the significance of this reaction, it's crucial to understand the role of hydrogen peroxide in different fields. In the environment, it helps in breaking down pollutants and improving water quality. In medicine, for example, hydrogen peroxide is used as a disinfectant due to its ability to kill bacteria and viruses. In laboratories, it is a key component in various chemical experiments and reactions.
Now, let's break down the balanced equation to see how the reaction unfolds. The equation indicates that two molecules of hydrogen peroxide react together. This is a classic example of a decomposition reaction, where a single compound breaks down into simpler substances Not complicated — just consistent..
- On the left side of the equation, we have two molecules of hydrogen peroxide.
- On the right side, we see the formation of two molecules of water and one molecule of oxygen gas.
This transformation is not only a matter of chemistry but also a demonstration of energy release. As hydrogen peroxide breaks down, it releases energy in the form of heat, which can be harnessed in various applications. Understanding this energy release is vital for optimizing reactions in industrial settings Not complicated — just consistent..
You'll probably want to bookmark this section.
When examining the decomposition process, don't forget to note the stability of hydrogen peroxide. Consider this: under normal conditions, hydrogen peroxide remains stable for a relatively long time. Even so, when exposed to heat or light, it can decompose rapidly. This aspect is crucial for applications that require controlled reactions, such as in the production of bleach or in laboratory settings where precise measurements are necessary That alone is useful..
The balanced equation also highlights the mole ratio involved in the reaction. In plain terms, for every two molecules of hydrogen peroxide, exactly two molecules of water and one molecule of oxygen are produced. This precise ratio is essential for chemists and scientists who need to predict the outcomes of their experiments accurately.
In practical terms, the decomposition of hydrogen peroxide has numerous implications. Take this case: in the field of water treatment, it is used to break down harmful substances, making water safer for consumption. By understanding the balanced equation, professionals can better manage the process, ensuring that the right amount of hydrogen peroxide is used to achieve the desired results Worth knowing..
Real talk — this step gets skipped all the time.
On top of that, this reaction is often used in educational settings to teach students about chemical equations and stoichiometry. That's why by working through the balanced equation, learners can develop a deeper understanding of how to balance chemical formulas and predict the products of reactions. This skill is not only academic but also highly applicable in real-world scenarios Not complicated — just consistent. Less friction, more output..
When discussing the decomposition of hydrogen peroxide, it's also worth mentioning its environmental impact. The release of oxygen gas is a critical aspect of this reaction. In nature, oxygen is essential for life, and understanding how it is produced through this process helps us appreciate the interconnectedness of chemical reactions and ecosystems.
In addition to its scientific significance, the balanced equation for hydrogen peroxide decomposition serves as a reminder of the importance of precision in chemistry. Every detail matters, whether it's the number of molecules involved or the conditions under which the reaction occurs. This precision is what allows scientists to innovate and improve processes across various industries.
As we explore the decomposition of hydrogen peroxide, we uncover a wealth of information about chemical behavior, energy dynamics, and practical applications. The balanced equation not only provides a clear representation of the reaction but also encourages a deeper appreciation for the complexities of chemistry. By mastering this concept, we equip ourselves with the knowledge to tackle more advanced topics and contribute meaningfully to scientific discussions.
At the end of the day, the decomposition of hydrogen peroxide is more than just a chemical equation; it is a gateway to understanding the broader implications of chemistry in our daily lives. On the flip side, by focusing on the balanced equation, we gain insight into the processes that shape our environment and enhance our understanding of scientific principles. Whether you are a student, educator, or simply a curious learner, delving into this topic can enrich your knowledge and inspire further exploration in the world of chemistry Surprisingly effective..
People argue about this. Here's where I land on it.
Another dimension that often goes unnoticed is the role of catalysts in modulating the rate at which hydrogen peroxide breaks down. While the simple balanced equation—2 H₂O₂ → 2 H₂O + O₂—captures the stoichiometry, it tells us nothing about how quickly the reaction proceeds under different conditions. In industrial settings, manganese dioxide (MnO₂), platinum, or even enzyme‑based catalysts such as catalase are introduced to accelerate the decomposition. Now, by lowering the activation energy, these catalysts enable the reaction to occur at ambient temperatures, making the process both energy‑efficient and safer. Understanding the interplay between catalyst choice, surface area, and reaction temperature can dramatically affect yields, especially when hydrogen peroxide is employed as a bleaching agent in paper manufacturing or as an oxidizer in rocket propulsion.
The thermodynamics of the reaction also merit attention. Think about it: engineers therefore design reactors with built‑in heat‑dissipation mechanisms, such as jacketed vessels or spray cooling, to maintain a stable temperature profile. Now, the decomposition of hydrogen peroxide is exothermic, releasing approximately –196 kJ mol⁻¹ of energy. This heat release can be harnessed intentionally—for example, in self‑heating food packs—or it can pose a hazard if the reaction proceeds uncontrolled. In laboratory demonstrations, the characteristic “foaming” of oxygen gas provides a vivid illustration of both the gas evolution and the accompanying temperature rise, reinforcing the concept that chemical change often involves concurrent physical changes Practical, not theoretical..
From a safety perspective, the balanced equation underscores the importance of stoichiometric control. An excess of hydrogen peroxide beyond the amount that can be safely decomposed may lead to over‑pressurization, especially in sealed containers. This is why regulations for the storage and transport of concentrated H₂O₂ specify limits on concentration (typically no more than 30 % for commercial use) and mandate vented containers. Also worth noting, because the reaction produces pure water and oxygen, the by‑products are benign—contrasting sharply with many other oxidizing agents that generate toxic or corrosive residues. This relative cleanliness makes hydrogen peroxide a favored oxidant in green chemistry initiatives, where the goal is to minimize waste and avoid hazardous by‑products Simple, but easy to overlook..
The ecological ramifications of the reaction are equally compelling. By injecting a controlled amount of H₂O₂ into a polluted zone, the generated hydroxyl radicals (·OH) attack organic contaminants, while the eventual decomposition yields oxygen that can support aerobic microbial degradation. Here's the thing — the balanced equation provides a baseline for calculating the required peroxide dose, ensuring that sufficient oxygen is produced without overwhelming the system with excess heat or pressure. In situ chemical oxidation (ISCO) techniques employ hydrogen peroxide to remediate contaminated soils and groundwater. Because of this, engineers can design treatment plans that are both effective and environmentally responsible.
Finally, the pedagogical value of the hydrogen peroxide decomposition extends beyond simple equation balancing. It serves as a gateway to several core concepts in chemistry curricula: redox reactions, reaction kinetics, thermochemistry, and catalysis. In practice, by conducting a series of experiments—varying catalyst type, temperature, or concentration—students can observe firsthand how each variable influences reaction rate and yield. Practically speaking, data collected from these experiments can be plotted to produce Arrhenius graphs, reinforcing the quantitative relationship between temperature and rate constants. Such hands‑on experiences cement abstract theory in tangible observation, fostering a deeper, more intuitive grasp of chemical principles That's the part that actually makes a difference..
Conclusion
The decomposition of hydrogen peroxide, succinctly expressed by the balanced equation 2 H₂O₂ → 2 H₂O + O₂, is a microcosm of chemical science in action. It intertwines stoichiometry with kinetics, thermodynamics with safety, and laboratory learning with real‑world applications ranging from water treatment to aerospace propulsion. By appreciating not only the numbers of atoms involved but also the conditions that govern the reaction’s speed, energy release, and environmental impact, we gain a holistic understanding that transcends the equation itself. This comprehensive perspective equips chemists, engineers, educators, and policymakers alike to harness the reaction responsibly, innovate responsibly, and teach effectively—ultimately reinforcing the principle that precise chemical knowledge is the foundation for sustainable progress Still holds up..