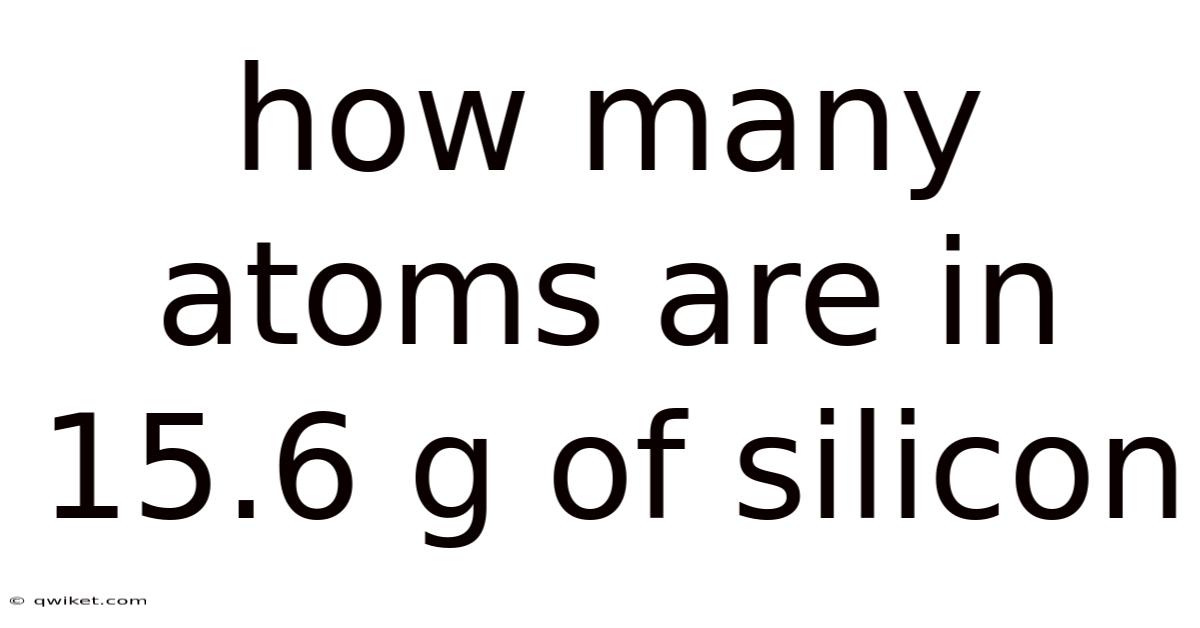Silicon is one of the most abundant elements on Earth and a cornerstone of modern technology, from computer chips to solar cells. Understanding how many atoms are present in a given mass of silicon is a fundamental exercise in chemistry that connects the macroscopic world we can weigh to the microscopic realm of individual atoms. This article explains, step by step, how to calculate the number of atoms in 15.6 g of silicon, explores the scientific principles behind the calculation, and answers common questions that often arise when working with moles, Avogadro’s number, and atomic mass No workaround needed..
Introduction: From Grams to Atoms
The moment you place 15.6 g of silicon on a balance, you are dealing with a bulk quantity that contains an astronomically large number of tiny particles. Converting that mass into a count of atoms requires three key pieces of information:
- The atomic mass of silicon (approximately 28.085 g mol⁻¹).
- The concept of the mole, which ties a mass to a specific number of entities.
- Avogadro’s constant (6.022 × 10²³ atoms mol⁻¹), the bridge between moles and individual atoms.
By applying these concepts, we can transform 15.6 g of silicon into a precise atom count, revealing just how densely packed matter truly is And it works..
Step‑by‑Step Calculation
1. Determine the molar mass of silicon
The periodic table lists silicon’s atomic weight as 28.085 g mol⁻¹. This value represents the mass of one mole of silicon atoms Still holds up..
2. Convert the given mass to moles
The number of moles (n) is obtained by dividing the sample mass (m) by the molar mass (M):
[ n = \frac{m}{M} = \frac{15.6\ \text{g}}{28.085\ \text{g mol}^{-1}} ]
Carrying out the division:
[ n \approx 0.555\ \text{mol} ]
(rounded to three significant figures, matching the precision of the given mass) It's one of those things that adds up..
3. Convert moles to atoms using Avogadro’s number
One mole contains 6.022 × 10²³ entities. Multiplying the mole amount by Avogadro’s constant (Nₐ) yields the atom count (N):
[ N = n \times N_{A} = 0.555\ \text{mol} \times 6.022 \times 10^{23}\ \text{atoms mol}^{-1} ]
[ N \approx 3.34 \times 10^{23}\ \text{atoms} ]
Thus, 15.6 g of silicon contains roughly 3.34 × 10²³ silicon atoms No workaround needed..
Scientific Explanation
The Mole Concept
The mole is a fundamental unit in chemistry that allows scientists to count particles without dealing with unwieldy numbers. In real terms, it is defined such that one mole of any substance contains exactly 6. 022 × 10²³ elementary entities—a value known as Avogadro’s number. This definition aligns the macroscopic scale (grams, liters) with the microscopic scale (atoms, molecules) Easy to understand, harder to ignore..
Why Atomic Mass Matters
Atomic mass reflects the average mass of an element’s isotopes as they occur naturally, expressed in atomic mass units (u). When converted to grams per mole, it tells us how much one mole of that element weighs. For silicon, the natural isotopic composition results in a weighted average of 28.085 g per mole. This value is crucial because it directly links the measurable mass (grams) to the abstract count of atoms (moles) And it works..
Avogadro’s Number in Practice
Avogadro’s constant is not just a large number; it embodies the bridge between the observable world and the quantum world. By multiplying the number of moles by Nₐ, we effectively “scale up” the count of entities from a manageable quantity (a mole) to the actual number of atoms present in the sample.
Significance of the Result
The calculated figure—3.34 × 10²³ atoms—is staggering. Worth adding: to put it in perspective, this is about half the number of grains of sand on a typical beach. Such comparisons highlight how dense matter is at the atomic level and why concepts like the mole are indispensable for chemists, material scientists, and engineers.
Practical Applications
Understanding atom counts is not merely an academic exercise. It underpins many real‑world processes:
- Semiconductor manufacturing: Precise doping of silicon wafers requires knowledge of how many impurity atoms are introduced per unit mass.
- Solar cell design: Efficiency calculations often start with the number of silicon atoms that can absorb photons.
- Materials budgeting: When ordering bulk silicon for industrial use, converting desired atom counts to mass ensures accurate procurement.
Frequently Asked Questions
Q1: Does the calculation change if the silicon is not pure?
A: The method remains the same, but you must first determine the mass fraction of silicon in the sample. Multiply the total mass by the purity percentage to obtain the mass of pure silicon, then apply the steps above Simple, but easy to overlook. Took long enough..
Q2: How accurate is the atomic mass value 28.085 g mol⁻¹?
A: The value is an average based on natural isotopic abundance and is accurate to at least four significant figures. For most laboratory calculations, this precision is sufficient. High‑precision work (e.g., isotope research) may require using specific isotopic masses Surprisingly effective..
Q3: Why do we round the final answer to three significant figures?
A: The input mass, 15.6 g, has three significant figures. According to the rules of significant‑figure arithmetic, the result should not be expressed with greater precision than the least precise measurement Simple, but easy to overlook..
Q4: Can I use a different unit, such as kilograms, in the calculation?
A: Yes. Convert the mass to kilograms (0.0156 kg) and the molar mass to kilograms per mole (0.028085 kg mol⁻¹). The ratio remains the same, yielding the identical mole value and ultimately the same atom count.
Q5: How does temperature affect the number of atoms?
A: Temperature does not change the number of atoms in a closed system; it only influences the kinetic energy and spacing between atoms. The mass‑to‑atom conversion is independent of temperature, assuming no phase change that alters the chemical composition Practical, not theoretical..
Common Mistakes to Avoid
- Confusing atomic mass with atomic number – The atomic number of silicon is 14 (number of protons), not its mass. Using 14 g mol⁻¹ would drastically underestimate the atom count.
- Neglecting significant figures – Reporting the result with too many digits suggests false precision. Match the precision of your inputs.
- Omitting Avogadro’s constant – Skipping the final multiplication step leaves you with moles, not atoms, which defeats the purpose of the calculation.
- Using the wrong unit for molar mass – Ensure the molar mass is expressed in grams per mole when the mass is in grams; otherwise, unit conversion errors will arise.
Real‑World Example: Doping a Silicon Wafer
Suppose a semiconductor engineer needs to introduce phosphorus atoms at a concentration of 1 × 10¹⁵ atoms cm⁻³ into a 300 µm‑thick silicon wafer with an area of 100 cm². First, calculate the wafer’s volume:
[ V = 100\ \text{cm}^2 \times 0.030\ \text{cm} = 3.0\ \text{cm}^3 ]
The desired number of dopant atoms is:
[ N_{\text{P}} = 1 \times 10^{15}\ \text{atoms cm}^{-3} \times 3.0\ \text{cm}^3 = 3 \times 10^{15}\ \text{atoms} ]
To determine the mass of phosphorus needed, convert atoms to moles (divide by Avogadro’s number) and then multiply by phosphorus’s molar mass (30.974 g mol⁻¹). This chain of conversions mirrors the silicon atom‑count calculation, illustrating why mastering the basic method is essential for advanced material engineering.
Conclusion
Calculating the number of atoms in 15.6 g of silicon is a straightforward yet powerful demonstration of how chemistry translates everyday measurements into the language of the atomic world. By:
- Using silicon’s molar mass (28.085 g mol⁻¹),
- Converting the sample mass to moles (≈ 0.555 mol), and
- Multiplying by Avogadro’s constant (6.022 × 10²³ atoms mol⁻¹),
we find that the sample contains approximately 3.34 × 10²³ silicon atoms. This figure not only satisfies academic curiosity but also serves as a foundational tool for fields ranging from semiconductor fabrication to renewable energy research. Mastery of the mole concept, atomic mass, and Avogadro’s number empowers students, scientists, and engineers to bridge the gap between the tangible mass on a balance and the invisible sea of atoms that constitute it.