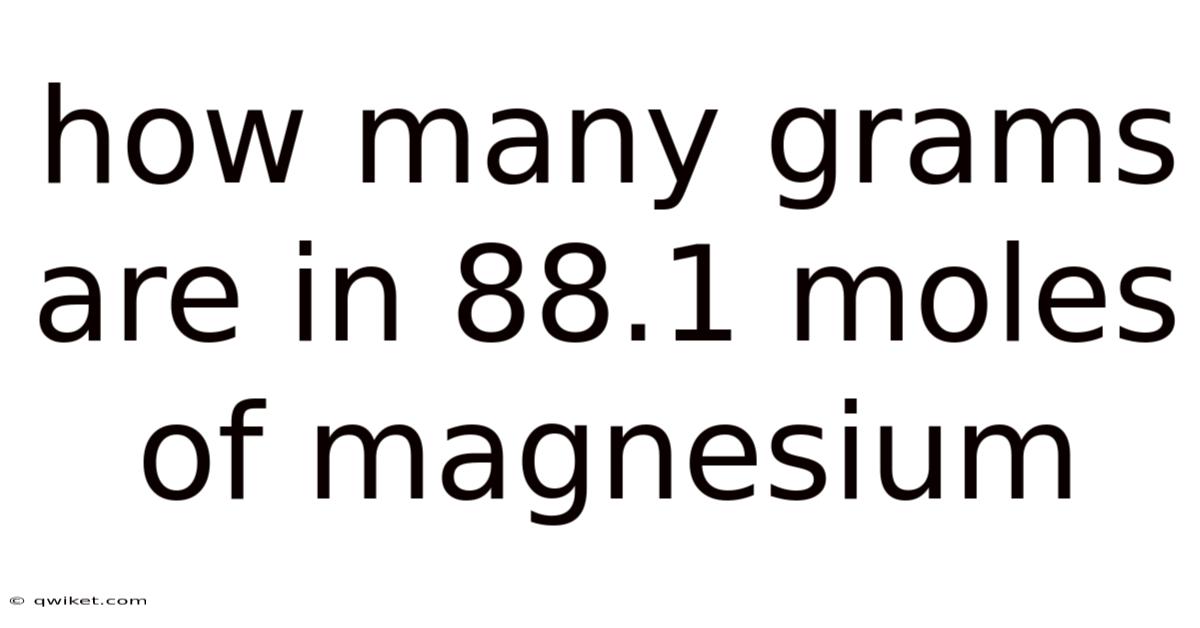The foundational principles guiding chemical quantification revolve around the precise relationship between atomic structure, molecular composition, and measurable quantities such as mass. But in the realm of chemistry, understanding how to translate moles into grams is a cornerstone skill that underpins countless experiments, industrial processes, and everyday scientific practices. This endeavor demands not only a grasp of basic stoichiometry but also an appreciation for the involved interplay between numerical values and physical realities. In real terms, at the heart of this process lies the concept of molar mass—a quantitative measure that links the atomic or molecular weight of a substance to its mass in grams per mole. That's why for instance, magnesium, a versatile metal celebrated for its role in construction, agriculture, and industrial applications, possesses a molar mass that dictates how its mass translates across different contexts. Recognizing this foundational concept allows practitioners to bridge abstract scientific principles with tangible results, ensuring that theoretical knowledge is effectively applied in practical scenarios. Think about it: whether determining the weight of a single crystal or calculating the total mass of a large-scale production batch, mastering this conversion is essential for precision and accuracy. The process itself involves several critical steps: identifying the substance in question, determining its atomic composition, calculating the molar mass, converting moles to grams, and finally verifying the calculations through cross-checking or real-world validation. Day to day, these stages collectively form a structured pathway that minimizes errors and enhances reliability, reinforcing the importance of meticulous attention to detail. In practice, in this context, the accuracy of the final result cannot be overstated, as even minor miscalculations can cascade into significant discrepancies when applied across multiple applications. On top of that, the application of such calculations extends beyond mere computation; they serve as a bridge between microscopic and macroscopic worlds, illustrating how fundamental concepts manifest in observable phenomena. This interconnectivity underscores why foundational knowledge remains indispensable in scientific inquiry, technological advancement, and problem-solving across disciplines. The process thus becomes a dynamic exercise where precision and understanding are inextricably linked, demanding both technical proficiency and a deep conceptual grasp to succeed effectively.
H2: Understanding Molar Mass and Its Significance
Molar mass serves as the linchpin connecting atomic-scale properties to observable mass. Beyond its utility in measurement, molar mass also influences the design of chemical reactions, as stoichiometric ratios often depend on the proportional relationships between substances. For magnesium, which belongs to the alkaline earth metal group, its atomic structure reveals a composition of approximately 24.In practical terms, knowing the molar mass allows scientists and engineers to predict how much substance will occupy a given volume or how much energy will be released or absorbed during reactions. So 3 grams, provides a concrete reference point for scaling quantities appropriately. It enables individuals to deal with the complexities of chemical systems with confidence, ensuring that their work remains grounded in well-established scientific principles. Thus, mastering the concept of molar mass is not merely an academic exercise but a practical necessity that underpins effective scientific practice. 3 atomic mass units per magnesium atom. Worth adding, the concept extends beyond individual calculations; it forms the basis for interpreting spectroscopic data, modeling chemical equilibria, and even designing synthetic compounds. 3 g/mol means that one mole of magnesium atoms weighs approximately 24.This understanding is particularly vital in fields such as pharmaceuticals, where precise dosages depend on accurate mass calculations, or in environmental science, where pollutant concentration assessments rely on similar principles. This is keyly the sum of the atomic weights of all atoms comprising a molecule or ion, providing a bridge between the microscopic world of atoms and the macroscopic world of measurable quantities like grams. The significance of molar mass cannot be overstated, as it acts as a universal constant that standardizes the conversion between grams and moles, ensuring consistency across diverse experimental settings. As an example, understanding that magnesium’s molar mass of around 24.On top of that, this value, derived from the periodic table, establishes the baseline for calculating its molar mass. In essence, molar mass acts as a universal translator, allowing the translation of atomic-level details into tangible outcomes that shape our understanding of the physical world. This foundational knowledge empowers professionals to approach challenges with a structured mindset, transforming abstract concepts into actionable insights that drive progress in countless domains.
H3: The Role of Molar Mass in Real-World Applications
The application of molar mass transcends theoretical applications and finds profound utility in numerous real-world scenarios, where its precise use can determine the success or failure of a project. In manufacturing industries, for instance, the production of magnesium alloys necessitates accurate control over composition, as deviations in molar mass can alter mechanical properties, thermal conductivity, or corrosion resistance. A manufacturer aiming to produce high-strength magnesium alloys must meticulously calculate the exact amount of magnesium required to achieve the desired alloy specifications, ensuring consistency and quality control. Similarly, in the field of agriculture, fertilizers formulated with magnesium compounds rely on precise stoichiometry to optimize nutrient release rates, directly impacting crop yields. In real terms, the agricultural sector’s reliance on such precision underscores the critical role of molar mass in ensuring that inputs deliver the intended benefits. Day to day, in the realm of education, educators use molar mass calculations to teach students fundamental principles of chemistry, fostering a deeper comprehension of atomic structure and molecular interactions. Also worth noting, the application extends to research and development, where experimental validation often hinges on accurate mass measurements. Here's a good example: when testing new catalysts for industrial processes, precise molar mass data is essential to confirm their efficacy and efficiency That's the part that actually makes a difference..
H4: Beyond the Lab: Molar Mass in Diverse Industries
The influence of molar mass isn’t confined to laboratories and academic settings; it’s a quietly vital component across a surprisingly broad spectrum of industries. Pharmaceutical companies, for example, apply molar mass calculations extensively during drug development. But determining the precise molar mass of a newly synthesized compound is critical for assessing its stability, predicting its behavior within the body, and ensuring accurate dosage calculations for patients. Similarly, in the food industry, molar mass plays a role in analyzing the nutritional content of food products. Determining the molar mass of vitamins, minerals, and other additives allows for precise labeling and accurate representation of a food’s composition. Forensic science also utilizes molar mass principles – particularly in analyzing trace evidence like unknown powders or residues – to identify substances and reconstruct events. Mass spectrometry, a technique heavily reliant on molar mass determination, provides crucial fingerprints for crime scene investigation.
Beyond that, the burgeoning field of materials science increasingly depends on molar mass considerations. Now, nanotechnology, with its focus on manipulating matter at the atomic scale, is perhaps the most acutely reliant on precise molar mass measurements – even slight variations can dramatically alter the behavior of nanomaterials. Think about it: designing new polymers, ceramics, and composites requires a thorough understanding of the molar mass of each component and its impact on the material’s final properties. Even seemingly unrelated sectors, like environmental monitoring, apply molar mass data to quantify pollutants and assess their impact on ecosystems Easy to understand, harder to ignore..
H5: Conclusion: A Cornerstone of Scientific Understanding
All in all, the seemingly simple concept of molar mass represents a cornerstone of scientific understanding and a critical tool across a multitude of disciplines. From the meticulous control of alloy production to the development of life-saving pharmaceuticals, its application is pervasive and profoundly impactful. It’s a testament to the power of fundamental scientific principles – a reminder that even the smallest unit of measurement, when understood and applied correctly, can tap into a wealth of knowledge and drive innovation. Mastering molar mass isn’t just about memorizing a formula; it’s about embracing a way of thinking that prioritizes precision, accuracy, and a deep appreciation for the relationships that govern the world around us.