The Name Of Fe2o3 Is ....
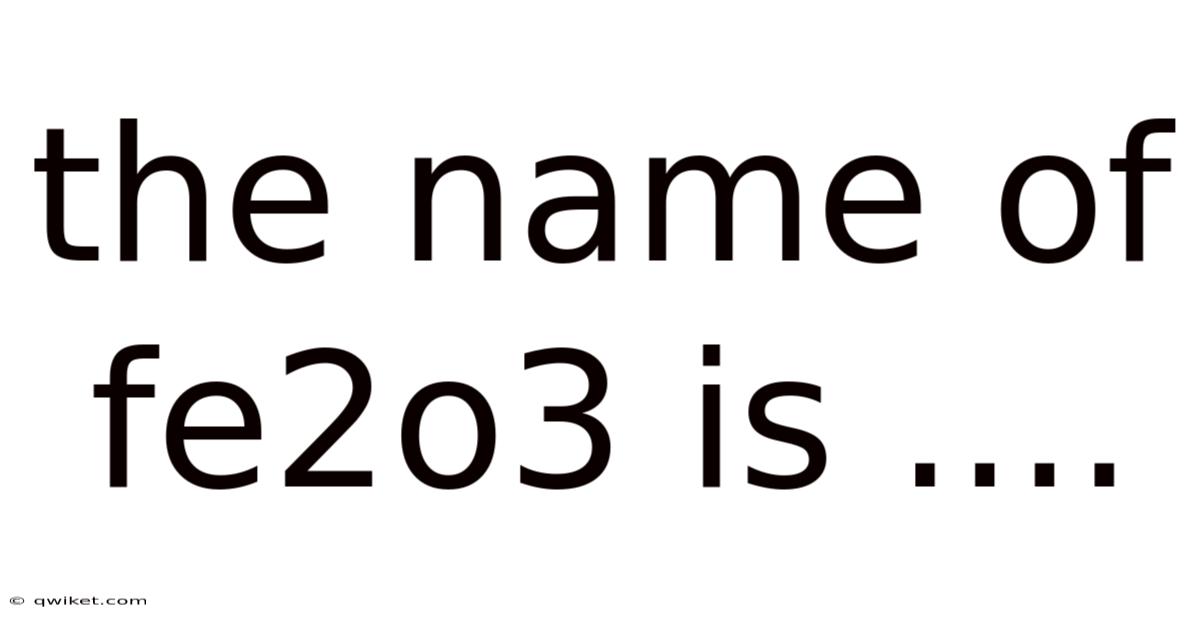
Understanding the name Fe₂O₃ is essential for anyone delving into the world of chemistry and material science. This compound, often recognized by its chemical formula, plays a significant role in various industrial and scientific applications. In this article, we will explore what Fe₂O₃ truly represents, why it is important, and how it impacts our daily lives. Let’s dive into the fascinating story behind this name and its implications.
When we look closely at the composition of Fe₂O₃, we see a fascinating interplay of elements. The term Fe₂O₃ stands for iron(III) oxide. This name is not just a label; it tells us a lot about the properties and uses of this compound. Let’s break it down to understand its significance.
The element iron is a vital component in many everyday materials. It is found in a variety of forms, but Fe₂O₃ is particularly noteworthy. This compound is commonly known as rust, which is a reddish-brown substance that forms when iron reacts with oxygen and water. Understanding the formation of Fe₂O₃ helps us appreciate its role in both natural and industrial processes.
Iron(III) oxide is characterized by its stability and resistance to corrosion, making it a valuable material in various applications. In the realm of chemistry, this compound is often used in the production of pigments, catalysts, and even in the manufacturing of ceramics. Its unique properties make it a subject of interest for scientists and engineers alike.
Now, let’s explore the reasons why Fe₂O₃ is more than just a chemical formula. The significance of this compound extends to several areas, including its applications in technology and environmental science. For instance, Fe₂O₃ is used in the production of batteries and other electronic devices, where its stability and conductivity play crucial roles.
Moreover, Fe₂O₃ has implications in the field of environmental protection. It can be utilized in the development of materials that help in reducing pollution. By understanding the properties of this compound, we can better appreciate its role in promoting sustainability. This connection between chemistry and environmental stewardship highlights the importance of studying Fe₂O₃ in depth.
To fully grasp the importance of Fe₂O₃, it’s essential to understand the steps involved in its formation. When iron reacts with oxygen in the presence of water, it undergoes a series of chemical transformations. This process leads to the creation of Fe₂O₃, which is not only stable but also has a wide range of applications. The steps of this reaction are crucial for scientists aiming to harness its potential effectively.
In addition to its chemical properties, Fe₂O₃ also has a significant impact on industries. It is commonly used in the manufacturing of paints and coatings, where its durability and resistance to weathering are highly valued. This application not only showcases its utility but also emphasizes the need for a deeper understanding of its composition and behavior.
As we explore the world of Fe₂O₃, it becomes clear that this compound is more than just a name on a chemical sheet. It is a gateway to understanding various scientific concepts and practical applications. By learning about the importance of Fe₂O₃, we can appreciate the intricate connections between chemistry and the world around us.
In conclusion, Fe₂O₃ is a compound that holds a special place in the field of chemistry. Its name, iron(III) oxide, encapsulates the essence of its properties and uses. Understanding this compound not only enhances our knowledge of chemical reactions but also opens doors to innovative applications in technology and environmental science. Whether you are a student, a professional, or simply a curious mind, delving into the details of Fe₂O₃ can inspire you to explore further and appreciate the beauty of science.
When examining the importance of Fe₂O₃, it’s crucial to recognize the multiple facets of its existence. From its chemical structure to its real-world applications, this compound serves as a reminder of the interconnectedness of science and daily life. By focusing on these aspects, we can foster a greater appreciation for the role of Fe₂O₃ in our modern world. Embrace the journey of learning about this remarkable compound, and let it ignite your curiosity about the wonders of chemistry.
Continuingseamlessly from the established themes of Fe₂O₃'s formation, properties, and significance:
Beyond its foundational role in corrosion and industrial applications, Fe₂O₃'s true potential often lies in its transformative capabilities. Its inherent catalytic properties, particularly when engineered at the nanoscale, unlock pathways for advanced environmental remediation. For instance, nanostructured Fe₂O₃ particles are actively researched for their ability to catalyze the breakdown of persistent organic pollutants and even facilitate the decomposition of greenhouse gases under specific conditions. This leverages the compound's redox activity, where iron atoms can readily switch between Fe³⁺ and Fe²⁺ states, enabling electron transfer reactions crucial for degradation processes. Furthermore, Fe₂O₃'s high surface area and porosity make it an excellent candidate for adsorbing heavy metal ions from contaminated water sources, acting as a natural filter material. This dual functionality – both catalytic and adsorptive – positions Fe₂O₃ as a cornerstone material in developing sustainable technologies aimed at purifying air and water, directly contributing to the environmental protection goals mentioned earlier.
The journey from understanding Fe₂O₃'s basic chemistry to harnessing its advanced applications underscores a fundamental truth: the study of fundamental materials is intrinsically linked to solving complex global challenges. Whether it's preventing the degradation of infrastructure, enhancing industrial processes, or developing cleaner technologies, Fe₂O₃ exemplifies how deep scientific inquiry into a seemingly simple compound can yield profound practical benefits. Its story is one of interconnectedness – between the atomic structure and macroscopic properties, between laboratory research and real-world impact, and between human ingenuity and the natural world.
In conclusion, Fe₂O₃, or iron(III) oxide, is far more than a common rust or a pigment. It is a testament to the elegance and utility embedded within inorganic chemistry. From its formation through oxidation to its diverse roles in industry, catalysis, and environmental science, this compound demonstrates the power of understanding material behavior. Its study not only deepens our comprehension of chemical principles but also actively contributes to building a more sustainable and technologically advanced society. Fe₂O₃ stands as a vital link between the foundational science of chemistry and the practical needs of our modern world, reminding us that the solutions to many contemporary problems often lie within the fundamental building blocks of matter.
Continuing the exploration of Fe₂O₃'s multifaceted nature, its journey from a simple oxide to a cornerstone of advanced materials science reveals a compelling narrative of innovation. Beyond its established roles in catalysis and adsorption, Fe₂O₃ is increasingly recognized as a vital component in next-generation energy technologies. Its inherent redox activity and abundance make it a prime candidate for electrochemical applications. Research is actively investigating nanostructured Fe₂O₃ and its composites as electrodes in rechargeable batteries, particularly for sodium-ion and potassium-ion systems, where its cost-effectiveness and stability offer promising alternatives to scarce or expensive materials. Furthermore, Fe₂O₃'s optical properties are being harnessed in emerging fields like photocatalysis for water splitting, aiming to generate hydrogen fuel sustainably, and in the development of novel sensors for detecting gases or biomolecules, leveraging its sensitivity to environmental changes.
This expanding horizon underscores a critical insight: Fe₂O₃'s true power lies not just in its individual properties, but in its remarkable versatility and adaptability. By strategically manipulating its structure – from nanoscale particles to complex heterostructures – and integrating it with other materials, scientists are unlocking unprecedented functionalities. This engineering approach transforms Fe₂O₃ from a passive material into an active participant in solving some of humanity's most pressing challenges: energy storage, clean water production, and environmental monitoring. It exemplifies the core principle of materials science: understanding fundamental properties allows us to design solutions for complex problems.
In conclusion, Fe₂O₃ stands as a powerful testament to the profound impact of fundamental chemical understanding. From its humble origins as rust or a pigment, it has evolved into a dynamic material central to environmental remediation, energy innovation, and advanced sensing. Its journey highlights the symbiotic relationship between scientific inquiry and practical application. By delving deep into the behavior of this seemingly simple compound, researchers have uncovered a wealth of potential, demonstrating that the solutions to contemporary global challenges often reside within the intricate dance of atoms and electrons that define inorganic chemistry. Fe₂O₃ is far more than an oxide; it is a versatile enabler, a bridge between the laboratory bench and the real-world needs of a sustainable and technologically advanced society, reminding us that the most impactful innovations often stem from mastering the fundamentals.
Latest Posts
Latest Posts
-
Objectivity In The Interpretation Of Data Is Referred To As
Mar 27, 2026
-
Signing Naturally Unit 4 Answer Key Pdf
Mar 27, 2026
-
04 04 A Close Look At Nature
Mar 27, 2026
-
What Is Poverty Jo Goodwin Parker
Mar 27, 2026
-
For The Purpose Of Calculating Gdp Investment Is Spending On
Mar 27, 2026